MICROBIOLOGY CULTURE INSTRUCTIONS Microbiology Programs
Inspect the fiberboard container carefully for all inclusions. After inventorying the contents of the container, store them in a refrigerator. These instructions are illustrated in the Program Guide online under Microbiology Instructions.
Special Safety Precautions - These specimens contain pathogens or potential pathogens and should be considered infectious and handled as though they are capable of transmitting disease. They should be handled and disposed of only by personnel trained to work with pathogenic bacteria. All laboratory precautions and safety measures appropriate to handling live cultures should be practiced when working with these specimens.
In addition to the Precautions section of the program guide, be especially careful to avoid aerosol creation, inhalation, ingestion or injection of bacteria. These specimens should be disposed of as hazardous waste.
Rehydration Instructions for Swab Samples – These specimens are stabilized viable microorganisms lyophilized directly to an inoculation swab. A Rehydration Fluid tube is provided for each swab. The Microbiology Rehydration Fluid is located with the rest of your samples and is not found inside the tube with the swabs. The Diluent vials are placed in a pouch labeled REHYDRATION FLUID. There are up to 5 equivalent vials, each containing approximately 0.5 mL of TSB. The vials are not specifically labeled for any sample or Microbiology program module. We recommend you label the rehydrating vial prior to use to avoid confusion.
a. Warm appropriate amount of media, one specimen swab and one vial of rehydrating fluid to room temperature (20-25ºC) for each specimen tested. Store the second swab and extra rehydrating fluid vials in the refrigerator in case repeat testing is necessary.
b. Remove swab from plastic tube and submerge the sample portion into the fluid. Allow approximately 10 seconds for the lyophilized specimen to liquify while mixing and swirling the swab gently.
c. Once the swab is saturated, inoculate your media directly with the swab. Return the swab to the rehydration fluid before inoculating each subsequent culture plate.
d. If you perform a gram stain as part of your identification procedure, you can now transfer a portion of the liquified specimen to a clean glass slide. Be sure to have inoculated all culture media prior to using the specimen swab to prepare your gram stain. NOTE: There will be a small amount of charcoal residue from the lyophilization process present on your gram stain slide. This should not interfere with your ability to determine the staining pattern of the organism.
e. Continue by following the procedure and methods used by your lab to identify the organism(s) present in these specimens.
For Uricult users or users of other tests that require a liquid sample.
a. Open each foil pouch at the tear slit and remove the swab from the foil pouch.
b. Submerge the swab portion only into the provided rehydration broth tube for 30 seconds to wet the swab.
c. Express as much material from the swab as possible by firmly pressing the head of the swab against the side of the rehydration broth tube.
d. Swirl the rehydration broth tube gently to ensure even mixing and then use the broth as you would a liquid sample.
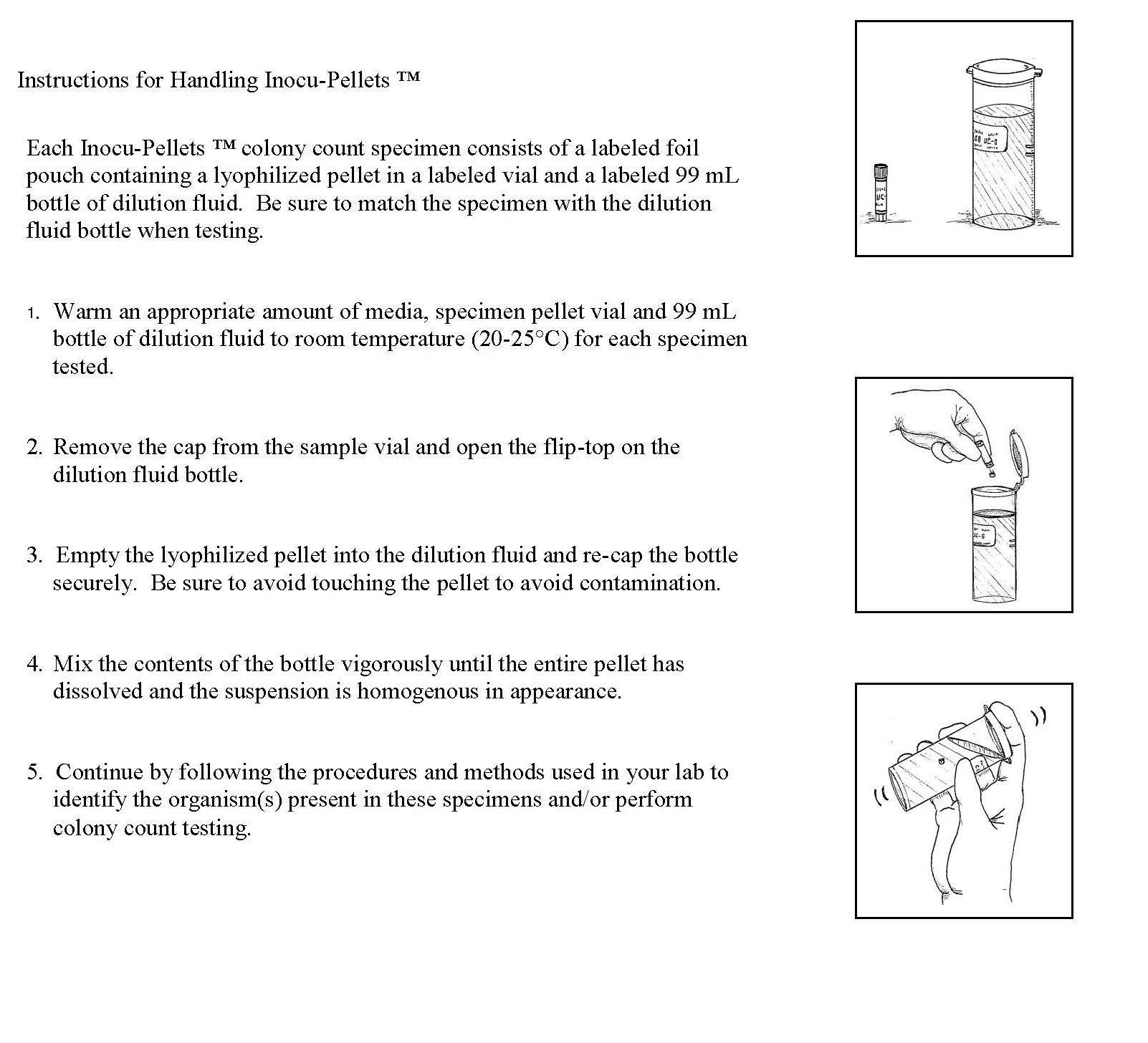
Urine Colony Count (CC-) Samples – Each sample consists of lyophilized pellet in a labeled foil pouch and a labeled 99 mL bottle of dilution fluid.
a. Warm an appropriate amount of media, specimen pellet and 99 mL bottle of dilution fluid to room temperature (20-25ºC) for each specimen tested.
b. Remove the cap from the sample vial and open the flip-top on the dilution fluid bottle.
c. Empty the lyophilized pellet into the dilution fluid and recap the bottle securely. Be careful to avoid touching the pellet to avoid contamination.
d. Mix the contents of the bottle vigorously until the entire pellet has dissolved and the suspension is homogeneous in appearance.
e.The entire contents of the dilution bottle (which now contains the dissolved lyophilized pellet) simulates a urine sample.
Proceed to test as you would a patient sample in your laboratory.
f. Continue by following the procedures and methods used by your lab to identify the organism(s) present in these specimens and/or perform colony count testing.
g. If you perform a gram stain as part of your identification procedure, you may transfer a small amount of the mixed solution from the first CC specimen to a clean glass slide. Be sure to use sterile technique when transferring the solution. NOTE: There will be a small amount of charcoal residue from the lyophilization process present on your glass slide. This should not interfere with your ability to determine the staining pattern of the organism.
h. Record your colony count results for only the first two CC specimens.
Reporting Results
Please review codes. Changes have been made to the organism codes.
|
766 |
Achromobacter sp. |
800 |
Fusobacterium sp. |
863 |
Serratia liquefaciens |
GENERAL RESULT CODES |
|
|
758 |
Achromobacter xylosoxidans |
804 |
Fusobacterium necrophorum |
865 |
Serratia marcescens |
943 |
Aerobe found, referred for ID |
|
738 |
Acinetobacter sp. |
801 |
Fusobacterium nucleatum |
866 |
Shigella sp. |
949 |
No aerobic growth |
|
740 |
Acinetobacter baumannii complex |
802 |
Gardnerella sp. |
870 |
Shigella boydii (Serotype C) |
947 |
No aerobic growth on blood agar |
|
739 |
Acinetobacter lwoffii |
803 |
Gardnerella vaginalis |
869 |
Shigella flexneri (Serotype B) |
944 |
Anaerobe found, referred for ID |
|
741 |
Aerococcus sp. |
828 |
Granulicatella sp. |
871 |
Shigella sonnei (Serotype D) |
946 |
Anaerobic cultures not performed - refer |
|
743 |
Aerococcus urinae |
808 |
Granulicatella adiacens |
873 |
Staphylococcus sp.; not S. aureus |
945 |
No anaerobes isolated |
|
742 |
Aerococcus viridans |
805 |
Haemophilus sp. |
874 |
Staphylococcus sp.; coagulase-negative |
951 |
No aerobic or anaerobic growth |
|
746 |
Aeromonas sp. |
806 |
Haemophilus haemolyticus |
875 |
Staphylococcus sp.; coagulase-positive |
|
|
|
747 |
Aeromonas hydrophila |
807 |
Haemophilus influenzae |
877 |
Staphylococcus aureus |
720 |
Gram negative bacilli |
|
717 |
Aeromonas hydrophila complex |
809 |
Haemophilus parahaemolyticus |
912 |
Staphylococcus capitis |
721 |
Gram negative coccobacilli |
|
703 |
Aggregatibacter sp. |
810 |
Haemophilus parainfluenzae |
878 |
Staphylococcus epidermidis |
722 |
Gram negative diplococci |
|
702 |
Aggregatibacter actinomycetemcomitans |
705 |
Kingella sp. |
880 |
Staphylococcus haemolyticus |
723 |
Gram positive bacilli |
|
701 |
Aggregatibacter aphrophilus |
704 |
Kingella kingae |
884 |
Staphylococcus hominis |
724 |
Gram positive cocci |
|
749 |
Alcaligenes sp. |
811 |
Klebsiella sp. |
872 |
Staphylococcus lugdunensis |
725 |
Gram positive diplococci |
|
748 |
Alcaligenes faecalis |
812 |
Klebsiella aerogenes |
900 |
Staphylococcus pseudintermedius |
941 |
No Salmonella or Shigella isolated |
|
752 |
Anaerococcus sp. |
813 |
Klebsiella oxytoca |
879 |
Staphylococcus saprophyticus |
930 |
No enteric pathogens isolated |
|
755 |
Anaerococcus prevotii |
814 |
Klebsiella pneumoniae |
890 |
Staphylococcus simulans |
948 |
No pathogens isolated |
|
750 |
Bacillus sp. |
707 |
Kocuria sp. |
867 |
Stenotrophomonas sp. |
718 |
Normal flora present |
|
753 |
Bacillus cereus |
708 |
Kocuria kristinae |
932 |
Stenotrophomonas maltophilia |
988 |
Contaminated Specimen |
|
756 |
Bacillus subtilis |
815 |
Lactobacillus sp. |
883 |
Streptococcus sp.; alpha-hemolytic |
960 |
Other organism not listed |
|
759 |
Bacteroides sp. |
820 |
Lacticaseibacillus casei |
889 |
Streptococcus sp.;beta-hemolytic; not Grp A |
|
|
|
855 |
Lacticaseibacillus sp. |
|
|
|
|
|
854 |
Lacticaseibacillus rhamnosus |
|
|
|
761 |
Bacteroides fragilis |
706 |
Laribacter hongkongensis |
892 |
Streptococcus sp. Group C - large colony |
|
760 |
Bacteroides fragilis group |
817 |
Listeria sp. |
893 |
Streptococcus sp. Group C - small colony |
|
744 |
Burkholderia sp. |
818 |
Listeria monocytogenes |
894 |
Streptococcus sp.; Group D |
|
714 |
Burkholderia cenocepacia |
819 |
Micrococcus sp. |
882 |
Streptococcus sp.; non-hemolytic |
|
745 |
Burkholderia cepacia |
821 |
Micrococcus luteus |
898 |
Streptococcus agalactiae |
|
715 |
Burkholderia cepacia complex |
751 |
Moellerella sp. |
794 |
Streptococcus anginosus |
|
716 |
Burkholderia gladioli |
754 |
Moellerella wisconsensis |
710 |
Streptococcus anginosus group |
|
763 |
Campylobacter sp. |
822 |
Moraxella sp. |
899 |
Streptococcus bovis group |
|
762 |
Campylobacter coli |
823 |
Moraxella catarrhalis |
885 |
Streptococcus constellatus |
|
764 |
Campylobacter fetus |
774 |
Moraxella lacunata |
903 |
Streptococcus dysgalactiae |
|
765 |
Campylobacter jejuni |
833 |
Moraxella osloensis |
891 |
Streptococcus gallolyticus |
|
767 |
Citrobacter sp. |
824 |
Morganella morganii |
895 |
Streptococcus intermedius |
|
768 |
Citrobacter braakii |
825 |
Neisseria sp. |
876 |
Streptococcus mitis group; not S. pneumo |
|
769 |
Citrobacter freundii |
856 |
Neisseria cinerea |
896 |
Streptococcus mutans group |
|
713 |
Citrobacter freundii complex |
826 |
Neisseria gonorrhoeae |
901 |
Streptococcus pneumoniae |
|
770 |
Citrobacter koseri |
829 |
Neisseria meningitidis |
887 |
Streptococcus pyogenes |
|
772 |
Clostridioides difficile |
832 |
Neisseria mucosa |
904 |
Streptococcus salivarius group |
|
771 |
Clostridium sp. |
827 |
Neisseria sicca |
902 |
Streptococcus sanguinis |
|
773 |
Clostridium perfringens |
788 |
Neisseria subflava |
897 |
Streptococcus viridans group |
|
775 |
Clostridium septicum |
836 |
Pantoea sp. |
905 |
Vibrio sp. |
|
777 |
Corynebacterium sp. |
839 |
Pantoea agglomerans |
908 |
Vibrio alginolyticus |
|
779 |
Corynebacterium jeikeium |
727 |
Parabacteroides sp. |
907 |
Vibrio parahaemolyticus |
|
782 |
Corynebacterium striatum |
726 |
Parabacteroides distasonis |
906 |
Vibrio vulnificus |
|
784 |
Corynebacterium ulcerans |
733 |
Pasteurella sp. |
909 |
Yersinia sp. |
|
783 |
Corynebacterium urealyticum |
732 |
Pasteurella multocida |
910 |
Yersinia enterocolitica |
|
785 |
Corynebacterium xerosis |
729 |
Pediococcus sp. |
709 |
Yersinia enterocolitica group |
|
781 |
Cronobacter sp. |
728 |
Pediococcus acidilactici |
911 |
Yersinia pseudotuberculosis |
|
780 |
Cronobacter sakazakii |
830 |
Peptostreptococcus sp. |
|
|
|
712 |
Cronobacter sakazakii group |
831 |
Peptostreptococcus anaerobius |
PRESUMPTIVE IDENTIFICATION CODES |
|
|
776 |
Cutibacterium sp. |
840 |
Plesiomonas sp. |
Genital Culture Codes |
|
|
786 |
Cutibacterium acnes |
924 |
Plesiomonas shigelloides |
722 |
Gram negative diplococci |
|
796 |
Eikenella sp. |
731 |
Prevotella sp. |
995 |
Gram negative diplococci absent |
|
790 |
Eikenella corrodens |
730 |
Prevotella bivia |
913 |
Negative for N. gonorrhoeae |
|
793 |
Elizabethkingia sp. |
834 |
Proteus sp. |
914 |
Positive for N. gonorrhoeae |
|
816 |
Elizabethkingia meningoseptica |
835 |
Proteus mirabilis |
915 |
Presumptive Positive for N. gonorrhoeae |
|
787 |
Enterobacter sp. |
837 |
Proteus vulgaris |
917 |
No growth on Thayer Martin |
|
789 |
Enterobacter cloacae |
847 |
Providencia sp. |
918 |
Growth on Thayer Martin, would refer |
|
711 |
Enterobacter cloacae complex |
843 |
Providencia rettgeri |
|
|
|
791 |
Enterococcus sp. |
848 |
Providencia stuartii |
Throat Culture Codes |
|
|
792 |
Enterococcus faecalis |
838 |
Pseudomonas sp. |
922 |
Negative for Group A Strep |
|
795 |
Enterococcus faecium |
841 |
Pseudomonas aeruginosa |
923 |
Positive for Group A Strep |
|
797 |
Enterococcus gallinarum |
842 |
Pseudomonas fluorescens |
920 |
Presumptive Positive for Group A Strep |
|
737 |
Erysipelothrix sp. |
844 |
Pseudomonas luteola |
|
|
|
736 |
Erysipelothrix rhusiopathiae |
845 |
Pseudomonas stutzeri |
Urine Culture Codes |
|
|
798 |
Escherichia sp. |
846 |
Salmonella sp. |
993 |
Presumptive gram-negative organism |
|
799 |
Escherichia coli |
858 |
Salmonella sp.; Group D |
994 |
Presumptive gram-positive organ |
|
888 |
Fannyhessea sp. |
850 |
Salmonella enterica spp. |
|
778 |
Fannyhessea vaginae |
852 |
Salmonella enteritidis |
|
735 |
Finegoldia sp. |
851 |
Salmonella typhi |
|
734 |
Finegoldia magna |
862 |
Serratia sp. |
Instructions for Handling INOCU-SWAB II™

Instructions for Handling INOCU-PELLETS